
Life Sciences Solution Brief
Egnyte helps Life Sciences teams collaborate securely, meet GxP and
ICH E6(R3) requirements, and always stay audit-ready.
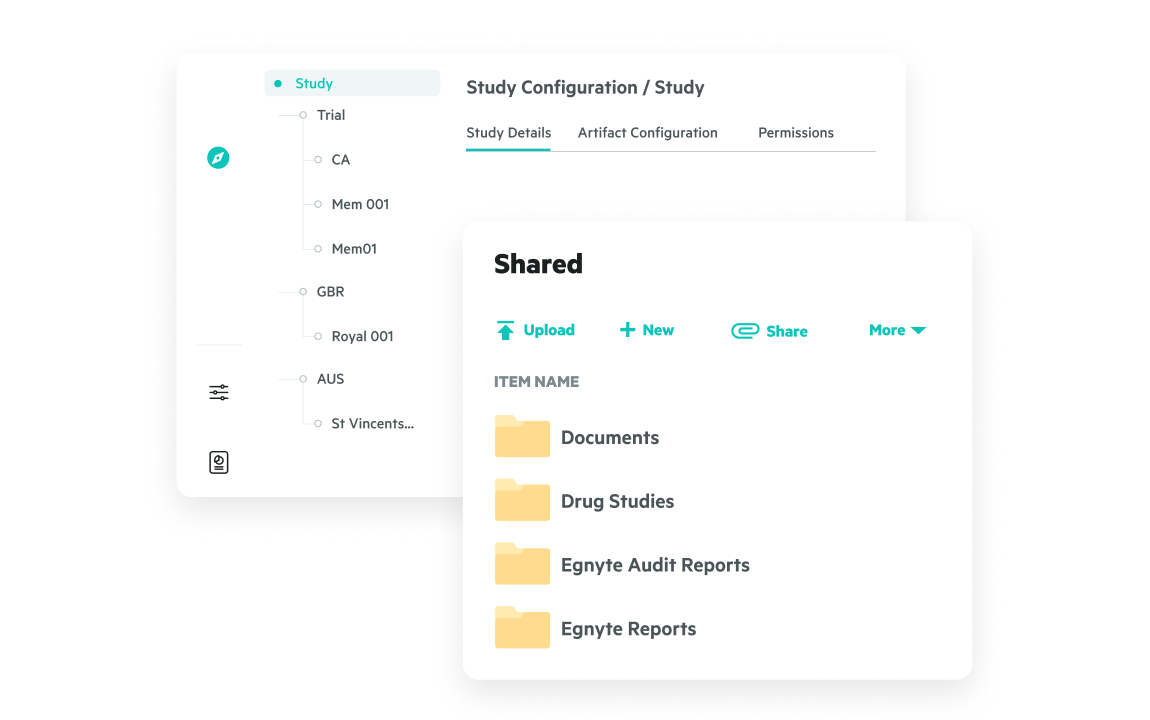
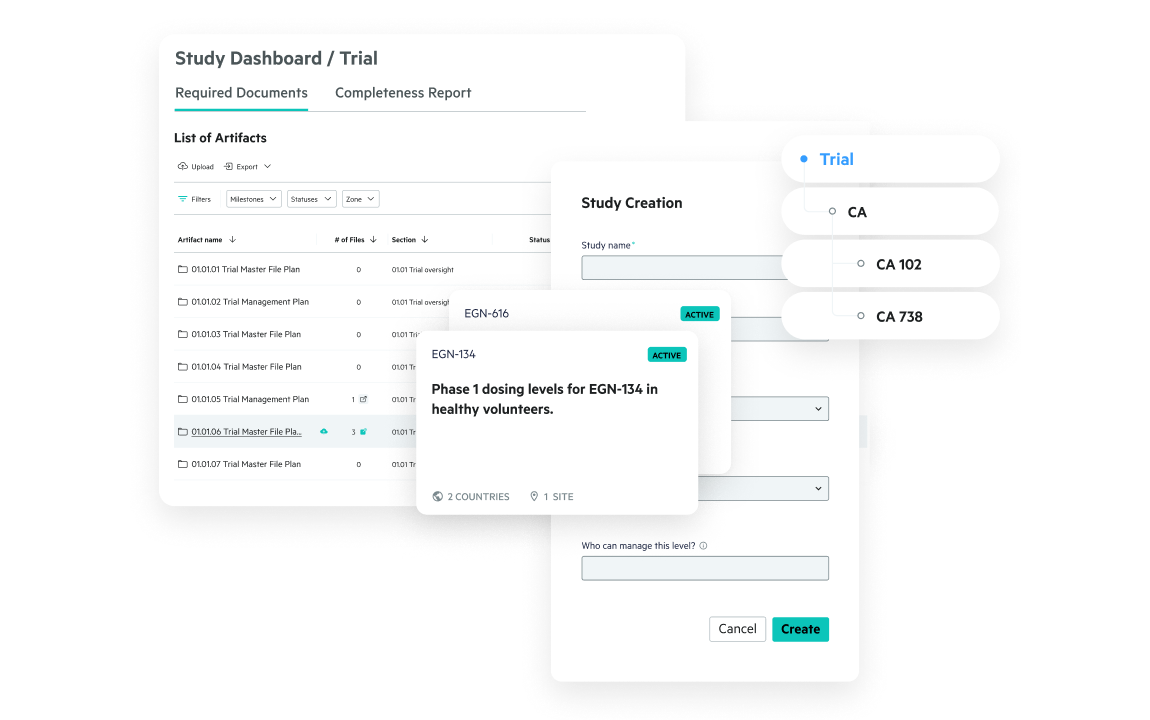
Centralize access to trial, quality, and R&D files—structured and unstructured—in a single secure repository. Eliminate silos, simplify collaboration, and improve visibility across your ecosystem.
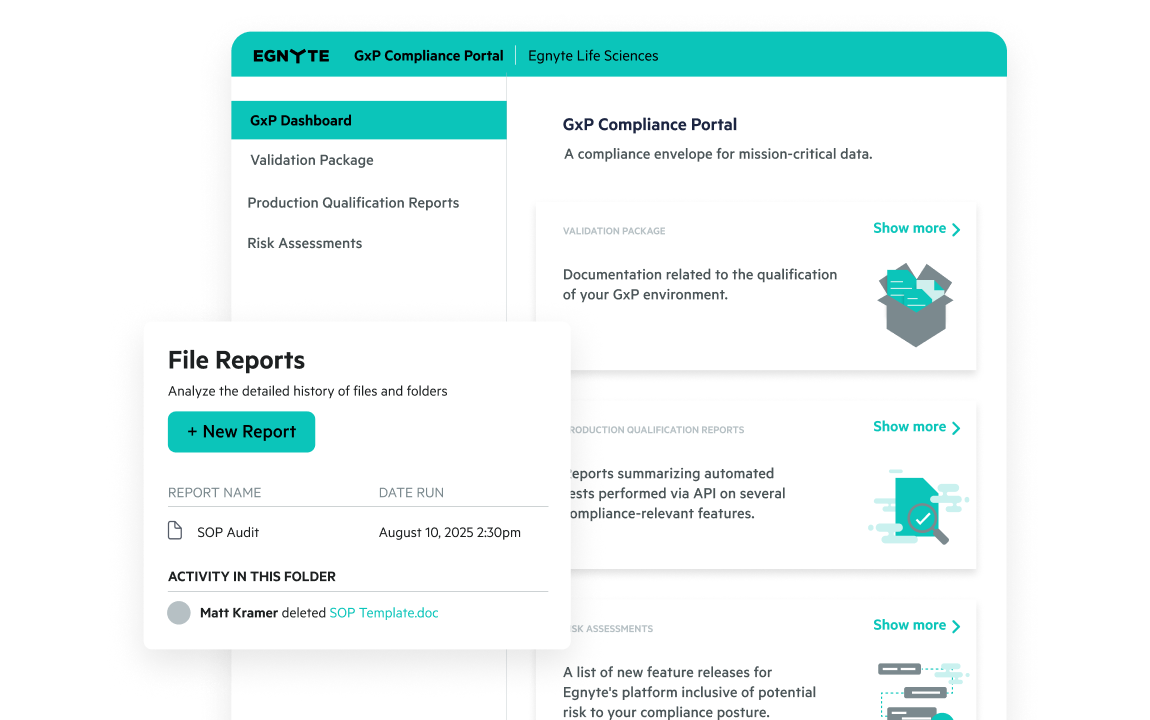
Support FDA, EMA, and global regulations with GxP-compliant audit trails, 21 CFR Part 11 e-signatures, and automated access controls. Reduce risk and ensure inspection readiness.
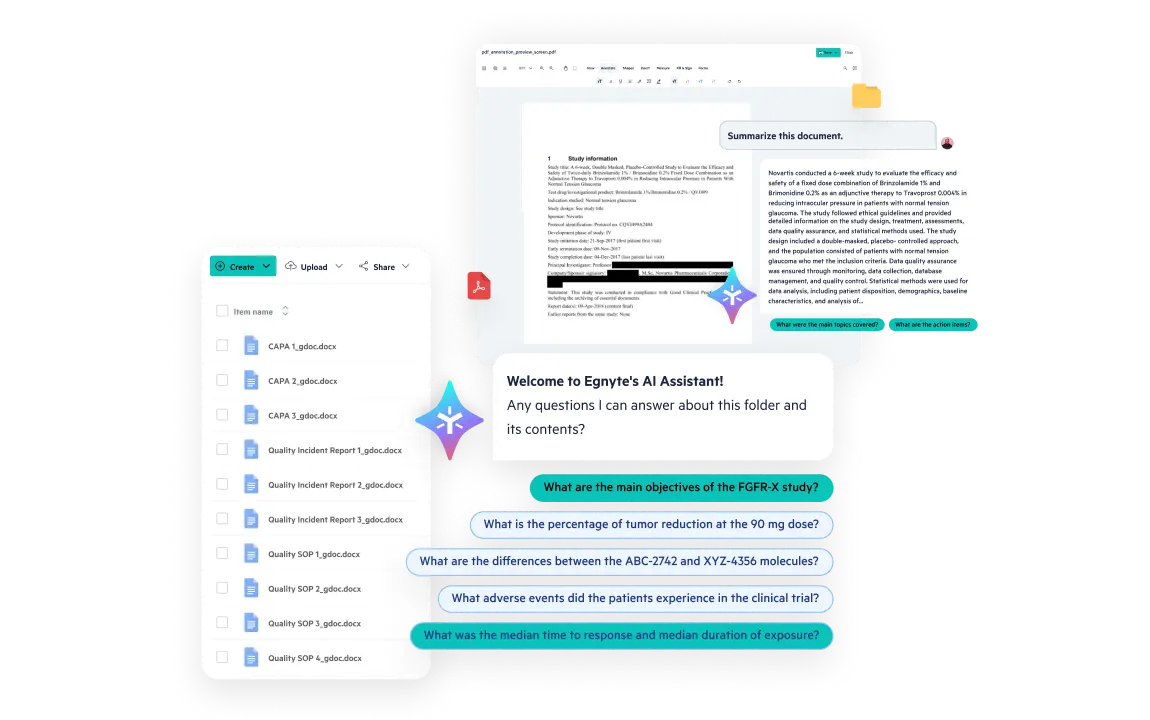
Leverage AI to tag metadata, summarize trial activity, and surface key insights across documents, CRFs, and protocols—using natural language queries.
Whether you're in preclinical, Phase III, or preparing for commercialization, Egnyte scales with your team’s needs and partner landscape.
Want to see what Egnyte has for your organization?
$22 / User / Month
Paid annually*
$39 / User / Month
Paid annually*
MOST POPULAR
Advanced AI
$48 / User / Month
Paid annually*
Advanced AI
Contact us for a
custom quote
Egnyte is trusted by thousands of IT leaders and end users. We are industry leaders in data security, data governance, cloud content collaboration, and tools for IT managers.
Cloud Content
Collaboration
Data
Security
Data
Governance
Data-Centric
Security
Sensitive
Data Discovery
Autodesk Construction
Cloud Integration
Partners
Electronic Trial
Master File (eTMF)
Have more questions? Contact Sales to get the answer you’re looking for.
Managing regulated data in the life sciences industry requires a secure, compliant, and scalable platform that ensures traceability of data across its lifecycle. The best solutions offer centralized data access, role-based permissions, and built-in support for FDA 21 CFR Part 11, Annex 11, GDPR, HIPAA and other regulatory standards. Validated platforms like Egnyte are commonly used to ensure the integrity, reliability, quality and security of both structured and unstructured data assets.
Ensuring a technology solution is compliant begins with understanding the intended use of the system and ensuring it is validated for that use. Solutions like Egnyte help streamline compliance by aligning platform requirements to regulatory requirements such as easy to access and comprehend audit trail reports and 21 CFR Part 11 compliant electronic signatures, as well as ensuring the platform is in a continuously validated state with inspection ready documentation available to all GxP customers.
Life sciences organizations manage unstructured data like lab reports, images, and research documents by using content management systems that provide scalable storage, advanced search, metadata tagging, and lifecycle management. Egnyte offers intelligent data governance and streamlined access to unstructured data for R&D, clinical, and regulatory teams.
Data security is critical in life sciences to protect sensitive research, intellectual property, clinical trial data, and patient information. Breaches can lead to regulatory fines and loss of trust. Secure platforms with encryption, access control, and real-time monitoring help safeguard data throughout the R&D lifecycle.
Cloud solutions enhance collaboration by enabling secure real-time access to research data, protocols, and regulatory documents across distributed teams. This improves efficiency, accelerates development timelines, and ensures that partners, CROs, and external stakeholders can easily and securely contribute to projects.
Key features to look for include regulatory compliance tools, secure data sharing, document version control, audit trails, automated workflows, and integrations with lab and clinical systems. A platform like Egnyte is tailored to meet the specific needs of biotech, pharma, and medical device companies.
From clinical trials to commercialization, Egnyte supports secure, scalable life sciences
workflows.