
3 Ways Egnyte Helps Life Sciences Companies Leverage the Cloud
Life sciences companies increasingly rely on cloud computing environments to accelerate research. The cloud provides cost effective compute power, more efficient data processing, access to files and applications from anywhere, and advanced analytics tools to gain insights from data and manage it.
But when a majority of that research is done by external contract research organizations (CROs) or in time zones on the other side of the world, that speed advantage can grind to a halt. And from a security standpoint, it’s suboptimal to give half a dozen CROs access to your Amazon Web Services (AWS) account.
In order to take full advantage of the power of public clouds like AWS and Microsoft Azure, life sciences companies must find solutions that allow a simple, secure, and efficient way to move data to and from the public cloud. They also need sufficient visibility and control over the data, all while maintaining the proper permission rights.
The Egnyte Public Cloud Connector
Egnyte’s Public Cloud Connector (PCC) makes it easy to connect your Egnyte environment to AWS and Azure, without having to write a single line of code. PCC syncs specific information from the Egnyte cloud into a preferred public cloud environment. It does so by automating the deployment of Egnyte’s hybrid device into a public cloud environment, which provides near real-time sync of files to and from Egnyte and the public cloud.
It also creates a point-in-time snapshot of information from a file system structure to an Azure blob or Amazon S3 bucket with a click of a button. This brings the files users interact with closer to the compute environment or to their workspace so employees can leverage PCC for a variety of use cases.
With Egnyte, life sciences companies get all the power of the public cloud combined with secure, real-time file access, sharing, and data syncing at a fraction of the cost.
3 Common Public Cloud Use Cases for Life Sciences
Life science firms are pairing Egnyte and the public cloud in myriad ways to get the most out of their research efforts, but here are three of the most common scenarios we see from our customers.
Scientific Processing Automation
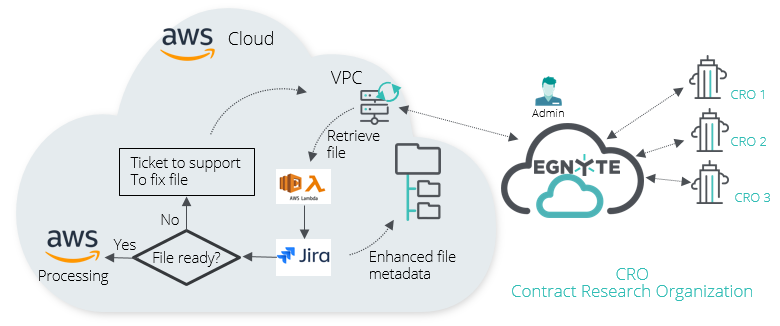
Example 1:
Customers use Egnyte to collect research data from external CROs. AWS can “grab” new files from specified folders through PCC and create a Jira ticket to notify the support team and track the progress of the research. The Jira ticket includes the link to the Egnyte source file, and the source file metadata is appended with a link to the Jira ticket.
AWS then validates the file. If the validation fails, the file is sent to support to be "fixed" and resubmitted. If it passes, AWS starts its cloud computing processing and puts the results back into Egnyte for analysis by the sponsor organization.
Without Egnyte, companies would need a much more manual and less secure workflow. That process could take weeks, requiring dedicated resources to manage and process the data. With Egnyte’s PCC, the process happens in near real-time.
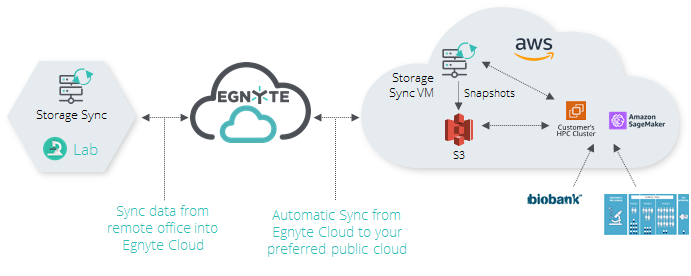
Example 2:
When customers generate microscopy imaging in a lab environment, such images require processing and integrations with databases such as a biobank. With the Egnyte on-premises Hybrid Device, teams can easily load the files directly from the imaging hardware to the hybrid device that will then sync collected information directly to the Egnyte cloud.
With PCC, customers can deploy data directly into their working environment in the cloud and sync files into that environment as well. The system can be set to automatically upload files from the remote office into the working environment in near-real-time. From there, customers’ files can be accessed by applications such as Amazon SageMaker and import information from the biobank database. Variants of DNA can be analyzed and the output can be visualized, all while maintaining the end-to-end permission access to the files.
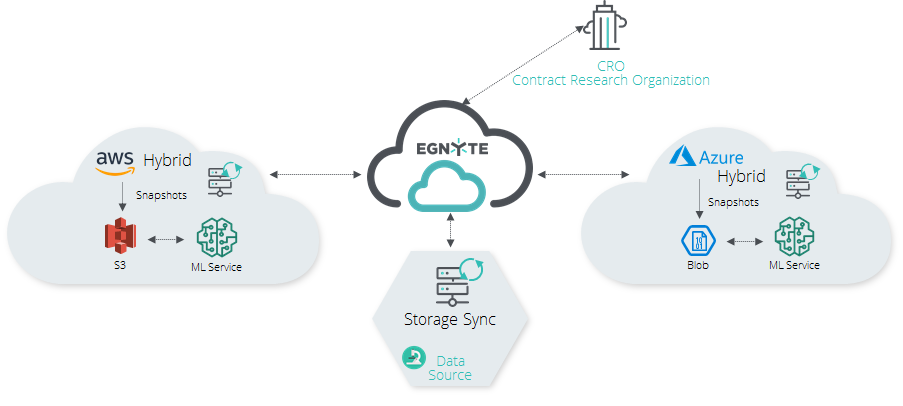
Simplified End-to-End Processes
Through a variety of lab devices such as microscopes, reams of imagery are generated. These large datasets make it difficult to migrate all the information from the source to where it can be sorted and processed while still maintaining the security and the permission rights to the data.
Public cloud computations allow a variety of options, from data sequencing to analysis of variants in DNA. Enabling this type of capability requires the data sources (video, imagery) to be ingested into a local server running Egnyte's on-premises Hybrid Device in the field. Files are then automatically uploaded into predefined folders on the Egnyte platform; from there it’s migrated to Azure or AWS.
With PCC, you can set up this connection in only a few clicks. This makes it simple to sync and transfer the data so it can be processed, then transferred back to the Egnyte cloud, where it is immediately available to all eligible users. Actions can be taken by users on any environment on Azure or AWS, and access to files before, during, or after processing can be shared with CROs. Such access is in the full control of the admin, who can revoke it at any time.
Information can also be synchronized between multiple environments so some teams can leverage AWS computation capabilities while others leverage Azure capabilities. PCC makes that data movement transparent to all involved teams.
In Egnyte for Life Sciences, your organization can manage the complete lifecycle of regulated documents with FDA 21 CFR Part 11-compliant e-signatures. Egnyte for Life Sciences Quality is a powerful tool that is tailor-made for emerging life sciences companies to meet FDA 21 CFR Part 11 regulations.
Our features enable an effective quality management process by facilitating collaboration, complying with regulatory requirements, and reducing the administrative burden of maintaining quality documents. All of these capabilities are available in a unified, easy to use, and audit-ready GxP-compliant platform.
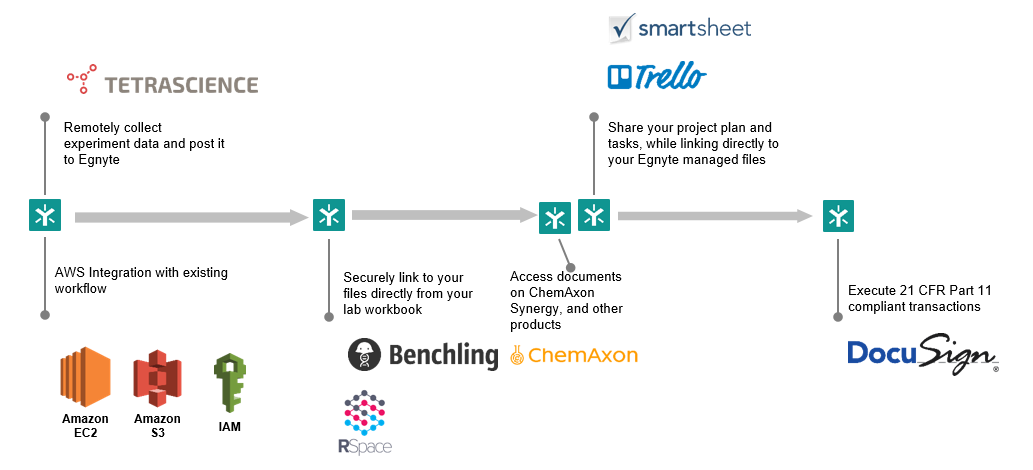
Ecosystem Support
Through their public APIs, AWS and Azure can integrate with countless third-party apps and services. This is a great way to add even more functionality to these powerful platforms, but it creates a management challenge for businesses.
Egnyte integrates with more than 150 industry specific applications, including Tetrascience, ChemAxon, Benchling, Rspace, Science Exchange, Catalytic, Dotmatics, xLM and ViQi. Life sciences companies get a single access point for all of their critical content in the cloud, which makes it easier to exchange data with external partners. Researchers can retrieve and organize trial data and handle large file formats, while reducing bandwidth usage at the same time.
IT teams can rely on this ecosystem support to centrally enforce consistent access across all repositories and all file types. They can also receive instant notifications when risks are detected, which makes it easier to act quickly when threats arise.
About Egnyte for Life Sciences
Egnyte for Life Sciences is a unified platform for life sciences documents and data. The advanced platform supports quality documentation, eTMFs, and features designed to maintain GxP compliance, securely share documents, and meet data privacy regulations. Customers benefit from managing regulated data, secure collaboration, and automated compliance with global data privacy laws. More than 16,000 businesses, and more than 600 life science organizations, trust Egnyte to provide visibility and control into their most valuable asset, their data. For more information, visit Egnyte for Life Sciences.




