Simplify GxP and 21 CFR Part 11 Compliance
- Leverage a 21 CFR Part 11 compliant environment aligned with GxP guidelines
- Minimize validation overhead via a predetermined release schedule
- Improve your controlled document management processes
* No Credit Card Required

Trusted by Over 600 Life Science Companies Worldwide

Egnyte Drives Excellence With 21 CFR Part 11 Compliance
Empower your organization with Egnyte's GxP-validated environment, ensuring adherence to 21 CFR Part 11 guidelines. Our platform simplifies the creation, storage, and management of GxP-regulated documents, offering native support for audit trails, data integrity, and robust access controls.
GxP and 21 CFR Part 11 Compliant Platform
- Easily create, store, and manage GxP-regulated documents
- Enjoy native support for audit trails
- Benefit from checksums for data integrity and robust access controls
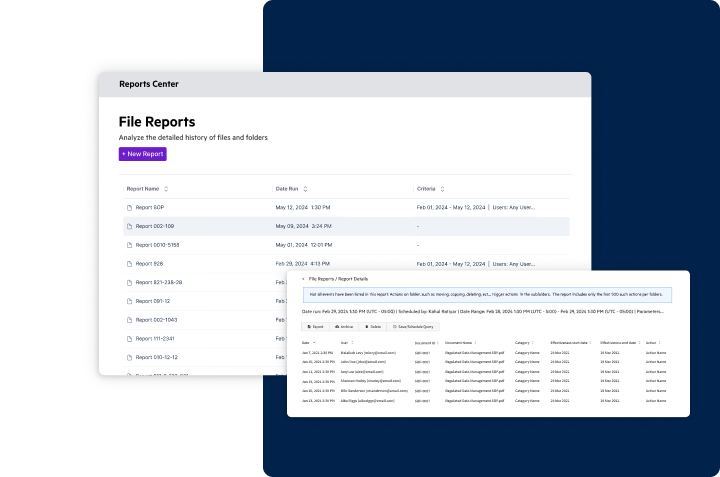
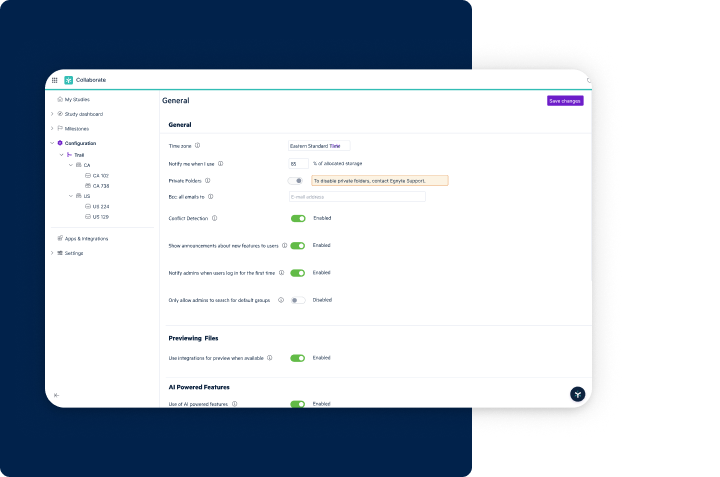
Easy Implementation & Management
- Establish a secure and regulated environment effortlessly
- Simplify auditing, validation, and reporting processes
- Tailor compliance to specific document types with user-friendly tools
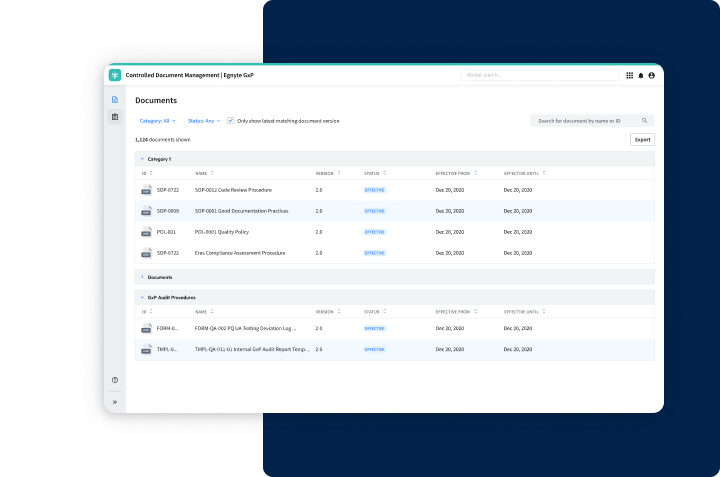
Improve Your Controlled Document Management Workflow
- Secure collaboration with CROs and partners
- Reduce administrative burdens associated with maintaining documentation
- Enhance the quality of your documentation process seamlessly
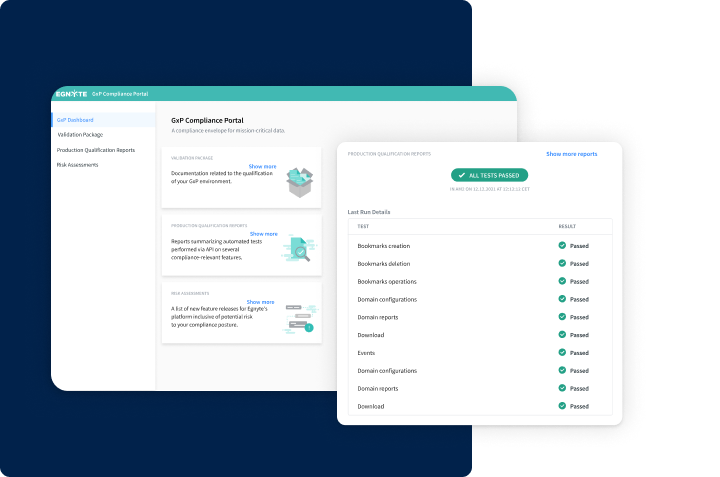
All-In-One Turnkey Platform
- GxP Compliance Portal
- Audit trails and rapid validation
- Content safeguards and ransomware detection and recovery
PC Mag
“Egnyte is one of the strongest offerings on the market.”
Gartner Peer Insights
“Gartner Peer Insights Customers’ Choice Winner for Content Collaboration Platforms”
G2
“A Leader in File Storage and Sharing for 2026”
Forrester
“390% Average Return on Investment (ROI) When Deploying Egnyte”
Discover The Capabilities Of Egnyte For Life Sciences
Start Free Trial **No credit card required. GxP available with the expanded Life Sciences GxP Plan.


