
Announcing Egnyte for Life Sciences Quality
The sheer number of parties involved in a clinical trial results in increased complexity. The ability to tame this complexity is what separates high-growth biotechs from the rest. In order to keep up with the pace of innovation organizations must improve efficiencies while adhering to regulatory requirements, all in a drive to maintain consistent data quality.
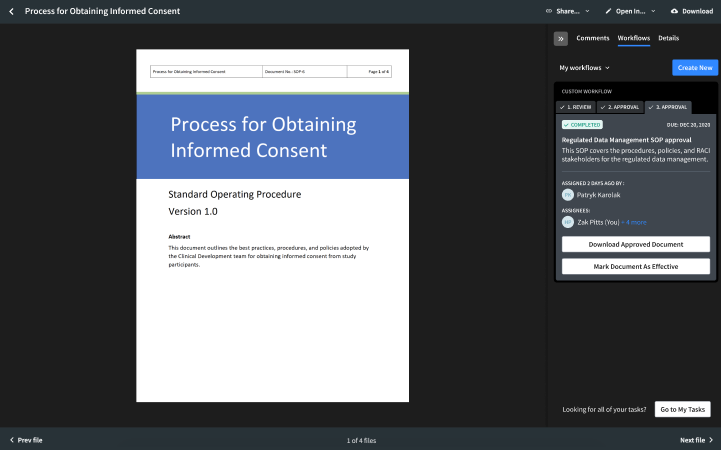
That is why we are introducing Egnyte for Life Sciences Quality, a new, powerful solution for life sciences companies designed to help them better manage regulated documentation. Egnyte for Life Sciences Quality is an easy-to-use, GxP-compliant solution that digitizes the lifecycle of regulated documents, like standard operating procedures (SOPs) and training records. With this product, Quality personnel can streamline review and approval workflows, and automate the handling of effective documentation, while meeting FDA 21 CFR Part 11.
Egnyte for Life Sciences Quality is designed to:
- Meet GxP & FDA 21 CFR Part 11. Our product digitizes regulated documents and workflows within a compliant software platform.
- Perform e-Signatures. Customers can sign off on documents, like SOPs and clinical trial protocols, in a compliant manner, without needing 3rd party plugins.
- Manage effective documents. As new versions are approved, Quality teams can easily define version numbers for new drafts, set expiry dates, and ensure only effective copies are available.
- Scale compliance processes quickly. Built with best practices gleaned from 16,000 global customers, the solution is easy to use, implement, and validate, as it integrates seamlessly with existing IT infrastructure.
- Support multiple departments and use cases. Designed with the ability to support training, continuity, and site readiness documentation.
Ready to see it in action? Watch the Egnyte for Life Sciences Quality Demo
A Unified Platform for Life Sciences Data and Documents
Egnyte for Life Sciences Quality further expands Egnyte’s offerings for life sciences organizations to manage Research, Development, and Business data on one unified platform. Our Quality solution provides a GxP-compliant environment that digitizes, centralizes, and manages deployment for regulated documents. With it, you get the best of both worlds, a robust system that enables the creation workflows, promotion and retirement of quality documentation—reducing administrative burden—and a central repository for effective documentation for end users—increasing compliance and adoption.
If you would like to learn more about Egnyte for Life Sciences Quality, reach out to a specialist.




